Call : 08045479613
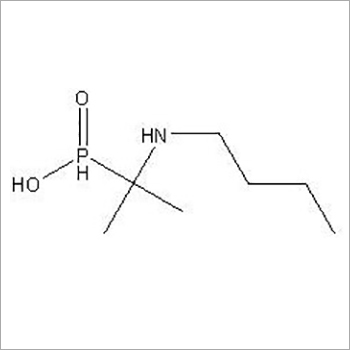
Butaphosphan api
Product Details:
- Molecular Weight 179.20 Grams (g)
- Molecular Formula C7H18NO2P
- CAS No 17316-67-5
- Click to View more
X
Butaphosphan api Price And Quantity
- 25 Kilograms
Butaphosphan api Product Specifications
- 179.20 Grams (g)
- C7H18NO2P
- 17316-67-5
Butaphosphan api Trade Information
- Cash Against Delivery (CAD) Cash on Delivery (COD) Letter of Credit (L/C) Letter of Credit at Sight (Sight L/C) Cash in Advance (CID) Cash Advance (CA)
- 2000 Kilograms Per Week
- 15 Week
- Yes
- Free samples are available
- 25 KGS
- All India
- GMP & ISO
Product Description
Enter Buying Requirement Details
Other Products in 'APIs Animal Health Drugs' category
 |
NGL FINE CHEM LTD.
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry


